A Mole of Any Compound Contains Avogadro's Number of
The amount of a substance that contains the same number of entities as there are atoms in 12 g of carbon-12. The Italian scientist Amedeo Avogadro reached the result that the mole of any substance contains a constant number of atoms molecules formula units or ions this number equals 6022140857 10 23 and it is called Avogadros number.
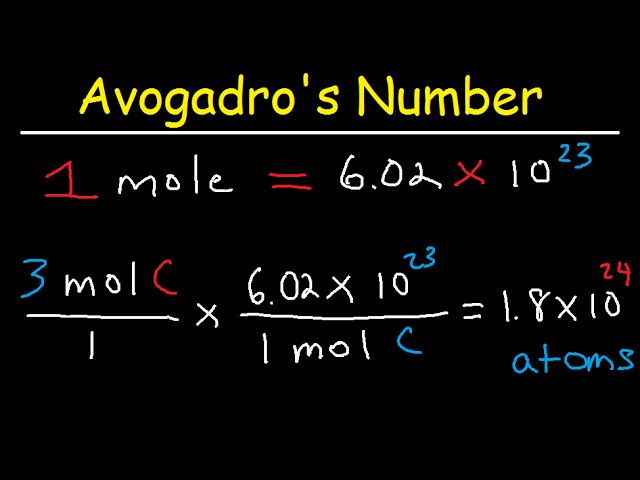
Avogadro S Number The Mole Grams Atoms Molar Mass Calculations Introduction Youtube
This very large number is called Avogadros Number.

. Chemists nonetheless have adopted the mole as their unofficial mascot right. A mole amount of substance contains the same number of particles as Avogadros Number. A mole of objects contains Avogadros number 6022 X 1023 objects.
One mole is the amount of any substance that contains the same number of units as the number of atoms in exactly 12 grams of carbon-12. One mole of any substance contains Avogadros number of particles. In basic words 602 l0 23 particles amount to one mole as twelve eggs are equal to one dozen.
Avogadros number is the number of the atoms the molecules formula units or ions which are found in one mole of. 1 mole of atoms 6022 10 23 atoms Gram atomic mass or Molar mass of element Number of moles frac Massquad ofquad element. One mole of NaCl contains 6022 x 10 23 NaCl formula units.
The mole or mol represents a certain number of objects. The number of units in one mole of any substance is called Avogadros number or Avogadros constant. So each element has different molar mass because on the different weights of 60221023 of their atoms.
A mole contains avogadros number of particles of a substance. A mole of iron atoms is 6022 X 1023 iron atoms. The units may be electrons ions atoms or molecules depending on the character of the reaction and the nature of the substance.
In this case a plausible statement would be. Since we will need to simplify moles to get atoms. One mole of H 2 O molecules contains 6022 x 10 23 molecules.
It is represented by No. A mole of any substance contains Avogadros number of representative particles or 602 x 1023 representative particles. 12044 x 10 23 atoms602210 23 atoms 2 moles.
1 mole of molecules 6022 10 23 molecules The number of particles present in 1 mole of any substance is fixed ie. A mole is the amount of any substance that has the same mass as Avogadros number of carbon-12 atoms. 1 mole contains 6022 x 10 23 entities Avogadros number.
It is equal to 602214085710 23. One mole of any substance has 6022x10²³ atoms molecules or ions Avogadros number into it. Just as a dozen apples is 12 apples a mole of apples is 6022 X 1023 apples.
602214 x 10 23 the number of units in a mole. Its value is 602210 23 atoms. Now the possible conversion factors could be.
A mole is the amount of a compound that has avogadros number of carbon-12 atoms in it. The mole or mol is a unit of measurement in chemistry used to designate a very large number of molecules atoms or particles. The NIST 2007 value of Avogadros number is 6022 141 79 0000 000 30 X 1023 mol1.
One mole of a substance is dependent on its molar mass. But here its given 12044 x 10 23 atoms thereby dividing it by Avogadros number. Hence the 602 l0 23 number of atoms molecules or A formula units is called Avogadros number that is equivalent to one mole of the respective compound.
A mole is the SI unit used to describe an amount of a substance. 6022 10 23. A mole of water molecules is 6022 X 1023 water molecules.
A mole is the amount of a compound that has Avogadros number of carbon-12 atoms in it. These numbers are very important for telling us about the quantity of elements. A mole is the amount of any substance that has the same mass as avogadros number of carbon-12 atoms.
The animal mole left is very different than the counting unit of the mole. It is equal to 6028 times 10 to the power 23 atoms or molecules of a substance. A mole contains Avogadros number of particles of a substance.
A mole of any substance contains Avogadros number 602 10 23 of representative particles. This number is called Avogadro constant or Avogadro number.

What Is The Relationship Between A Mole And Avogadro S Number A Plus Topper

The Mole Concept Avogadro S Number X Ppt Download

Lecture 5 The Mole Avogadro S Number The Mole Is Used When We Re Talking About Numbers Of Atoms And Molecules Tiny Particles Moleatomsmolecules The Ppt Download
No comments for "A Mole of Any Compound Contains Avogadro's Number of"
Post a Comment